Highlights
- •Amivantamab–chemotherapy improved PFS and intracranial PFS versus chemotherapy.
- •Amivantamab–lazertinib–chemotherapy improved PFS and intracranial PFS versus chemotherapy.
- •Predominant AEs in the amivantamab-containing arms were hematologic, EGFR, and MET related.
- •MARIPOSA-2 is the first study to demonstrate improved PFS versus chemotherapy after disease progression on osimertinib.
Background
Amivantamab plus carboplatin–pemetrexed (chemotherapy) with and without lazertinib demonstrated antitumor activity in patients with refractory epidermal growth factor receptor (EGFR)-mutated advanced non-small-cell lung cancer (NSCLC) in phase I studies. These combinations were evaluated in a global phase III trial.
Patients and methods
A total of 657 patients with EGFR-mutated (exon 19 deletions or L858R) locally advanced or metastatic NSCLC after disease progression on osimertinib were randomized 2 : 2 : 1 to receive amivantamab–lazertinib–chemotherapy, chemotherapy, or amivantamab–chemotherapy. The dual primary endpoints were progression-free survival (PFS) of amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy. During the study, hematologic toxicities observed in the amivantamab–lazertinib–chemotherapy arm necessitated a regimen change to start lazertinib after carboplatin completion.
Results
All baseline characteristics were well balanced across the three arms, including by history of brain metastases and prior brain radiation. PFS was significantly longer for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy [hazard ratio (HR) for disease progression or death 0.48 and 0.44, respectively; P < 0.001 for both; median of 6.3 and 8.3 versus 4.2 months, respectively]. Consistent PFS results were seen by investigator assessment (HR for disease progression or death 0.41 and 0.38 for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy, respectively; P < 0.001 for both; median of 8.2 and 8.3 versus 4.2 months, respectively). Objective response rate was significantly higher for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy (64% and 63% versus 36%, respectively; P < 0.001 for both). Median intracranial PFS was 12.5 and 12.8 versus 8.3 months for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy (HR for intracranial disease progression or death 0.55 and 0.58, respectively). Predominant adverse events (AEs) in the amivantamab-containing regimens were hematologic, EGFR-, and MET-related toxicities. Amivantamab–chemotherapy had lower rates of hematologic AEs than amivantamab–lazertinib–chemotherapy.
Conclusions
Amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy improved PFS and intracranial PFS versus chemotherapy in a population with limited options after disease progression on osimertinib. Longer follow-up is needed for the modified amivantamab–lazertinib–chemotherapy regimen.

Key words
Introduction
Mutations in the epidermal growth factor receptor (EGFR) gene are the most common actionable genomic alterations in non-small-cell lung cancer (NSCLC). Of EGFR mutations, 85%-90% are exon 19 deletions (Ex19del) and L858R substitution mutations.1,2 The current first-line standard of care in EGFR-mutated NSCLC is the third-generation EGFR tyrosine kinase inhibitor (TKI) osimertinib,3,4 which improved progression-free survival (PFS) and overall survival versus first-generation TKIs.5,6 Despite initial efficacy, nearly all patients treated with osimertinib develop resistance.7 Mechanisms of resistance to osimertinib are diverse and polyclonal, with the most common being alterations in the MET gene (e.g. up to 51% by fluorescence in situ hybridization)8 and EGFR pathways.9, 10, 11, 12 Guidelines recommend platinum-based chemotherapy as the next line of therapy,13,14 with a historical median PFS of 4.4-5.5 months in patients with disease progression after TKI treatment.15, 16, 17, 18Amivantamab, an EGFR-MET bispecific antibody with immune cell–directing activity,19, 20, 21 is approved for the treatment of patients with EGFR exon 20 insertion mutations whose disease progressed on or after platinum-based chemotherapy.22 Amivantamab has multiple mechanisms of action, including ligand blocking, receptor degradation, and engagement of effector cells (natural killer cells, monocytes, and macrophages) via its optimized Fc domain.19,20 Mechanistically, by binding extracellularly, amivantamab bypasses intracellular mutations (including those at the TKI catalytic domain), and its bispecific nature addresses MET as a mechanism of resistance. Clinically, amivantamab has shown activity against a wide range of activating and resistance mutations in EGFR-mutated NSCLC and in patients with MET exon 14 skip mutations.23, 24, 25, 26Lazertinib is a highly selective, central nervous system (CNS)–penetrant, third-generation TKI with demonstrated efficacy in activating EGFR mutations and T790M resistance.27,28 Simultaneously targeting the extracellular and catalytic EGFR domains by combining amivantamab with lazertinib (amivantamab–lazertinib) has been shown to provide a synergistic benefit.29,30 Amivantamab–lazertinib has demonstrated clinically meaningful activity in patients with EGFR-mutated NSCLC after disease progression on osimertinib.31 Additionally, continuation of a CNS-penetrant, third-generation TKI, such as lazertinib, after disease progression on osimertinib has been thought to be important since brain metastasis is a frequent outcome for patients with EGFR-mutated NSCLC.32The addition of carboplatin–pemetrexed (chemotherapy) to amivantamab or amivantamab plus lazertinib could address osimertinib-based resistance. In a phase I study, amivantamab–chemotherapy demonstrated an objective response rate of 44% in a safety population of 20 patients with advanced and refractory NSCLC.33 In a separate phase I study, amivantamab–lazertinib–chemotherapy was evaluated in 20 patients with EGFR-mutated NSCLC whose disease had progressed on prior TKIs and showed an objective response rate of 50%.34 The aforementioned results supported further evaluation.MARIPOSA-2 is a global, randomized, phase III trial assessing the efficacy and safety of amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy in patients with EGFR-mutated advanced NSCLC whose disease had progressed on or after osimertinib monotherapy.
Patients and methods
Patients
MARIPOSA-2 (ClinicalTrials.gov identifier, NCT04988295) enrolled patients who were 18 years of age or older and had locally advanced or metastatic EGFR-mutated (Ex19del or L858R) NSCLC with disease progression on or after osimertinib monotherapy (as the most recent line of treatment). Patients with brain metastases were eligible provided intracranial disease was clinically stable, asymptomatic, and on stable doses of steroids; prior definitive treatment with radiation or surgery was not required. For additional criteria, see Supplementary Methods, available at https://doi.org/10.1016/j.annonc.2023.10.117.
Trial oversight
The trial was conducted in accordance with the provisions of the Declaration of Helsinki, Good Clinical Practice guidelines (as defined by the International Conference on Harmonisation), applicable regulatory requirements, and the policy on bioethics and human biologic samples of Janssen Pharmaceuticals (trial sponsor). Each patient (or legally acceptable representative) provided written consent for participation. Informed consent could be obtained remotely by telephone or video conferencing where permitted by local regulations.MARIPOSA-2 was designed by the sponsor, who was responsible for the collection and analysis of the data and interpreted the trial data in collaboration with the authors. The first draft of the manuscript was written by the authors, with medical writing support funded by the sponsor and conducted in accordance with Good Publication Practice guidelines. All authors made the decision to publish and vouch for data completeness and accuracy, data analyses, and adherence to the clinical trial to the protocol. The protocol, amendments, and statistical analysis plan are available in the Supplementary Material, available at https://doi.org/10.1016/j.annonc.2023.10.117.
Trial design and treatment
Patients were randomly assigned in a 2 : 2 : 1 ratio to receive amivantamab–lazertinib–chemotherapy, chemotherapy alone, or amivantamab–chemotherapy in 21-day cycles (Supplementary Figure S1, available at https://doi.org/10.1016/j.annonc.2023.10.117). Amivantamab was administered intravenously at 1400 mg (1750 mg for body weight ≥80 kg) weekly for the first 4 weeks, and then 1750 mg (2100 mg for body weight ≥80 kg) every 3 weeks starting at cycle 3 (week 7). The first amivantamab infusion was split over 2 days, with 350 mg on cycle 1, day 1 and the remainder on cycle 1, day 2. Lazertinib was administered orally at 240 mg daily. Chemotherapy was administered intravenously at the beginning of every cycle, with pemetrexed at 500 mg/m2 administered every cycle and carboplatin at area under the curve 5 for the first four cycles. Amivantamab, lazertinib, and pemetrexed treatments were to be continued until disease progression or lack of clinical benefit as deemed by the investigator. Treatment blinding was not possible due to differences in administration, premedication requirements, and safety profiles of the regimens. Randomization was stratified by osimertinib line of therapy (first or second), race (Asian or non-Asian), and history of brain metastasis (yes or no).Originally, the MARIPOSA-2 study design included amivantamab–chemotherapy for the purposes of establishing contribution of components between amivantamab–lazertinib–chemotherapy and chemotherapy. In addition, there was a prespecified biomarker analysis with hypothesis testing, which was to be based upon next-generation sequencing (NGS). Emerging data did not validate the proposed NGS biomarker,26,31 so the analysis plan needed to be updated. Due to emerging phase I data that demonstrated promising antitumor activity of amivantamab–chemotherapy,33 the study was amended to allow dual hypothesis testing to independently assess amivantamab–chemotherapy versus chemotherapy. This decision was thus based on factors external to the MARIPOSA-2 study; no interim analysis of safety or efficacy was carried out before the implementation of the modification.Based on reports of increased rates of venous thromboembolism (VTE) for amivantamab plus lazertinib, which were identified midway through the trial,31,35 oral and subcutaneous anticoagulants (using either a direct oral anticoagulant or low-molecular-weight heparin, consistent with local guidelines) were recommended but not mandatory for patients receiving amivantamab–lazertinib–chemotherapy for the first 4 months of amivantamab–lazertinib treatment.During the study, the independent data monitoring committee observed increased hematologic and gastrointestinal toxicities occurring within the first four cycles of the amivantamab–lazertinib–chemotherapy arm. The original dosing schedule for amivantamab–lazertinib–chemotherapy was modified to defer the start of lazertinib until after completion of carboplatin. Due to limited follow-up after the regimen change and to further describe the safety and efficacy of the modified regimen, a separate open-label, randomized extension cohort comparing the modified amivantamab–lazertinib–chemotherapy regimen versus amivantamab–chemotherapy is ongoing and will enroll an additional 90 patients (Supplementary Figure S2, available at https://doi.org/10.1016/j.annonc.2023.10.117). All amendments are described in the protocol.
Endpoints
The dual primary endpoints evaluated PFS as determined by blinded independent central review according to Response Evaluation Criteria in Solid Tumors version 1.136 for amivantamab–chemotherapy versus chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy. As per protocol, all patients in the amivantamab–lazertinib–chemotherapy arm, regardless of the dosing schedule, were compared to those receiving chemotherapy. Secondary endpoints included objective response rate, duration of response, overall survival, PFS after first subsequent therapy, symptomatic PFS, intracranial PFS, and safety. All endpoints are listed and defined in the protocol.
Trial assessments
Disease assessments occurred at baseline, at 6 (+1) weeks after randomization, then every 6 (±1) weeks for the first 12 months, and then every 12 (±1) weeks thereafter until disease progression was confirmed by blinded independent central review. Brain magnetic resonance imaging was carried out at baseline, 6 (+1) weeks, 12 (±1) weeks, and then every 12 (±1) weeks until intracranial disease progression confirmed by blinded independent central review. Baseline assessments were carried out within 28 days before randomization. Adverse events (AEs), vital signs, and laboratory tests were graded using the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. Electrocardiograms were assessed at baseline.
Statistical analysis
The efficacy analysis included all randomly assigned patients on an intent-to-treat basis, while the safety analysis included all randomized patients who received at least one dose of any study treatment. For PFS, it was estimated that 600 patients with 350 events in all three arms combined would provide approximately 83% and 93% power for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy, respectively, versus chemotherapy to achieve a statistically significant difference for a hazard ratio (HR) of 0.65, with an overall two-sided α of 0.05. This sample size determination assumed a median PFS of 5.5 months for chemotherapy15,16 and 8.5 months for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy.Multiplicity was adjusted using a graphical approach. For dual hypothesis testing, treatment effects of amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy were independently compared to chemotherapy based on a log-rank test stratified by line of therapy, history of brain metastases, and Asian race, with initial testing of PFS at a two-sided α of 0.03 and 0.02, respectively. If both were significant, based on the prespecified α recycling procedure, objective response rate was then evaluated at 0.0267 and 0.0233 for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy, respectively, versus chemotherapy. If objective response rate was significant, overall survival was then evaluated for its first interim analysis via O’Brien Fleming α spending approach implemented by Lan–DeMets method. An interim overall survival analysis was planned at the time of primary analysis for PFS. Assuming PFS and objective response were significant for both amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy, then the interim analysis of overall survival would be evaluated at α of 0.000197 and 0.000138, respectively.For PFS and overall survival, HRs and 95% confidence intervals (CIs) were calculated from a stratified Cox regression model with treatment as the sole explanatory variable, using the same stratification factors as for the log-rank test. Median and corresponding 95% CIs were estimated by the Kaplan–Meier method. Objective response was analyzed using a stratified logistic regression model with treatment as the explanatory variable.Analyses of the additional secondary or other outcomes including the subgroup analyses, which were not part of the hypothesis testing in the study, are reported as point estimates and 95% CIs without adjusting for multiplicity. Additional statistical and multiplicity details are provided in the Supplementary Appendix, available at https://doi.org/10.1016/j.annonc.2023.10.117.All data reported here are based on the primary analysis. The data cut-off date was 10 July 2023.
Results
Patients and treatment
From December 2021 to April 2023, a total of 970 patients were screened and 657 patients were randomized (131 to amivantamab–chemotherapy, 263 to amivantamab–lazertinib–chemotherapy, and 263 to chemotherapy). One patient in the amivantamab–chemotherapy arm, none in the amivantamab–lazertinib–chemotherapy arm, and 20 in the chemotherapy arm did not receive treatment, leaving 636 (97%) patients who were treated (Supplementary Figure S3, available at https://doi.org/10.1016/j.annonc.2023.10.117). Demographics and baseline disease characteristics were well balanced across all three arms (Table 1). The percentage of patients with a history of brain metastases and the percentage of these who had received prior radiation treatment to the brain were also similar between arms. At a median follow-up of 8.7 months, the median treatment duration was 6.3 months (range 0-14.7 months) for amivantamab–chemotherapy, 5.8 months (range 0.1-18.6 months) for amivantamab–lazertinib–chemotherapy, and 3.7 months (range 0-15.9 months) for chemotherapy (Supplementary Table S1, available at https://doi.org/10.1016/j.annonc.2023.10.117). The most common reason for treatment discontinuation (of all agents) was progressive disease, at 41 (32%), 68 (26%), and 152 (63%) patients in the amivantamab–chemotherapy, amivantamab–lazertinib–chemotherapy, and chemotherapy arms, respectively.
Table 1Demographics and baseline disease characteristics
| Characteristic | Chemotherapy (n = 263) | Amivantamab–chemotherapy (n = 131) | Amivantamab–lazertinib–chemotherapy (n = 263) |
| Age | |||
| Median (range), years | 62 (31-85) | 62 (36-84) | 61 (23-83) |
| Category, n (%) | |||
| <65 years | 166 (63) | 79 (60) | 163 (62) |
| ≥65 years | 97 (37) | 52 (40) | 100 (38) |
| Sex, n (%) | |||
| Female | 157 (60) | 81 (62) | 168 (64) |
| Male | 106 (40) | 50 (38) | 95 (36) |
| Race, n (%) | |||
| Asian | 127 (48) | 63 (48) | 125 (48) |
| White | 123 (47) | 60 (46) | 129 (49) |
| Othera | 13 (5) | 8 (6) | 9 (3) |
| Region of enrollment, n (%) | |||
| Asiab | 126 (48) | 67 (51) | 131 (50) |
| Europec | 96 (37) | 45 (34) | 96 (37) |
| North America | 22 (8) | 13 (10) | 21 (8) |
| South America | 19 (7) | 6 (5) | 15 (6) |
| Body weight, kg | |||
| Median (range) | 63 (37-118) | 63 (39-112) | 64 (35-118) |
| Category, n (%) | |||
| <80 kg | 226 (86) | 113 (86) | 226 (86) |
| ≥80 kg | 37 (14) | 18 (14) | 37 (14) |
| ECOG performance status, n (%) | |||
| 0 | 101 (38) | 55 (42) | 92 (35) |
| 1 | 162 (62) | 76 (58) | 171 (65) |
| History of smoking, n (%) | |||
| No | 168 (64) | 90 (69) | 175 (67) |
| Yes | 95 (36) | 41 (31) | 87 (33) |
| Unknown | 0 | 0 | 1 (0.4) |
| Time from metastatic diagnosis, median (range), months | 21.0 (0.1-99.1) | 23.0 (0.2-115.3) | 21.5 (0.9-115.3) |
| Histologic type, n (%) | |||
| Adenocarcinoma | 260 (99) | 130 (99) | 260 (99) |
| Otherd | 3 (1) | 1 (1) | 3 (1) |
| History of brain metastases, n (%) | 120 (46) | 58 (44) | 120 (46) |
| No prior brain radiation | 61 of 120 (51) | 24 of 58 (41) | 56 of 120 (47) |
| Type of EGFR mutation, n (%) | |||
| Exon 19 deletion | 183 (70) | 89 (68) | 165 (63) |
| Exon 21 L858R | 79 (30) | 42 (32) | 98 (37) |
| Previous osimertinib line of therapy,e n (%) | |||
| Osimertinib as first line | 181 (69) | 97 (74) | 185 (70) |
| Osimertinib as second line | 82 (31) | 34 (26) | 77 (29) |
Those with a history of smoking were defined as patients who used tobacco, cigarettes, cigars, or pipes.
ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor.
a Other includes Black or African American, American Indian or Alaska Native, multiple, unknown, and not reported.
b Turkey counted as part of Asia.
c Russia counted as part of Europe.
d Other includes large cell carcinoma, squamous cell carcinoma, and other.
e One patient in the amivantamab–lazertinib–chemotherapy arm received osimertinib later than second line and is not included in the table.
In the amivantamab–lazertinib–chemotherapy arm, 166 patients received lazertinib concurrently with all other agents and 97 received lazertinib after completion of carboplatin, with median follow-up of 11.5 and 5.4 months, respectively. As a result, the majority of patients receiving the modified amivantamab–lazertinib–chemotherapy regimen (lazertinib after completion of carboplatin) have had very limited treatment with lazertinib at the time of data cutoff. Data on efficacy and safety of the modified regimen will be presented in a future publication.
Efficacy
The median PFS by blinded independent central review was 6.3 months (95% CI 5.6-8.4 months) for patients treated with amivantamab–chemotherapy, 8.3 months (95% CI 6.8-9.1 months) with amivantamab–lazertinib–chemotherapy, and 4.2 months (95% CI 4.0-4.4 months) with chemotherapy (Figure 1A). PFS was significantly longer in the amivantamab–chemotherapy arm compared to the chemotherapy arm (HR for disease progression or death 0.48, 95% CI 0.36-0.64, P < 0.001) and in the amivantamab–lazertinib–chemotherapy arm compared to the chemotherapy arm (HR for disease progression or death 0.44, 95% CI 0.35-0.56, P < 0.001).
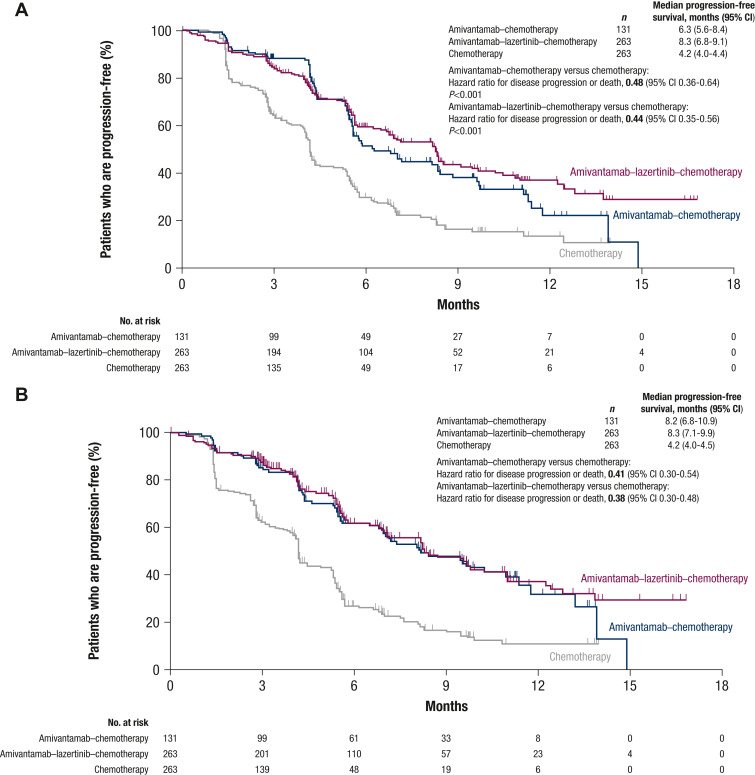
Figure 1 Progression-free survival by blinded independent central review and by investigator.
Shown are Kaplan–Meier estimates of progression-free survival assessed by blinded independent central review (A) and investigator assessment (B). The efficacy analysis set included all randomized patients. Tick marks indicate censoring of data. CI, confidence interval.
The median investigator-assessed PFS was 8.2 months (95% CI 6.8-10.9 months) for amivantamab–chemotherapy and 8.3 months (95% CI 7.1-9.9 months) for amivantamab–lazertinib–chemotherapy versus 4.2 months (95% CI 4.0-4.5 months) for chemotherapy, corresponding to HRs for disease progression or death of 0.41 (95% CI 0.30-0.54) and 0.38 (95% CI 0.30-0.48), respectively (Figure 1B and Supplementary Figure S4 and Table S2, available at https://doi.org/10.1016/j.annonc.2023.10.117).
The PFS benefit was consistent across predefined subgroups for amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy (Figure 2), including by history of brain metastases (Supplementary Figure S5, available at https://doi.org/10.1016/j.annonc.2023.10.117), osimertinib line of therapy (Supplementary Figure S6, available at https://doi.org/10.1016/j.annonc.2023.10.117), and EGFR mutation type (Supplementary Figure S7, available at https://doi.org/10.1016/j.annonc.2023.10.117).
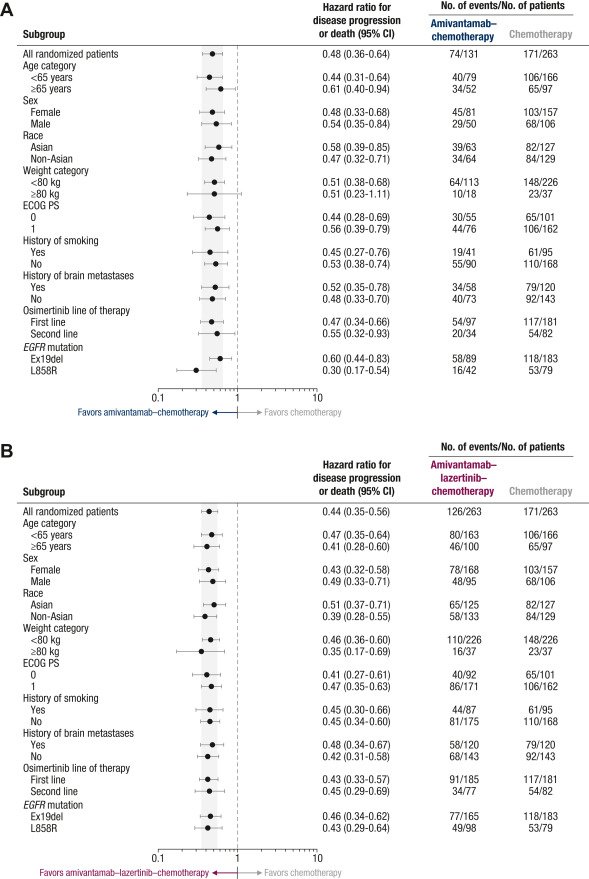
Figure 2 Progression-free survival by blinded independent central review of patient subgroups.
Shown are forest plots of progression-free survival in patient subgroups assessed by blinded independent central review for amivantamab–chemotherapy versus chemotherapy (A) and for amivantamab–lazertinib–chemotherapy versus chemotherapy (B). The efficacy analysis set included all randomized patients. The shaded areas indicate the 95% CI for the overall hazard ratio (all patients). CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status.
The objective response rate was 64% (95% CI 55%-72%) for amivantamab–chemotherapy, 63% (95% CI 57%-69%) for amivantamab–lazertinib–chemotherapy, and 36% (95% CI 30%-42%) for chemotherapy (Table 2; Supplementary Figure S8, available at https://doi.org/10.1016/j.annonc.2023.10.117), with significant improvements versus chemotherapy for amivantamab–chemotherapy (odds ratio 3.10, 95% CI 2.00-4.80, P < 0.001) and amivantamab–lazertinib–chemotherapy (odds ratio 2.97, 95% CI 2.08-4.24, P < 0.001). Among confirmed responders, median duration of response was 6.9 months (95% CI 5.5 months-not estimable) for amivantamab–chemotherapy, 9.4 months (95% CI 6.9 months-not estimable) for amivantamab–lazertinib–chemotherapy, and 5.6 months (95% CI 4.2-9.6 months) for chemotherapy.
Table 2Key efficacy endpoints by blinded independent central review
| Endpoint | Chemotherapy (n = 263) | Amivantamab–chemotherapy (n = 131) | Amivantamab–lazertinib–chemotherapy (n = 263) |
| Progression-free survival | |||
| No. of months, median (95% CI) | 4.2 (4.0-4.4) | 6.3 (5.6-8.4) | 8.3 (6.8-9.1) |
| % of patients progression-free at 6 months (95% CI) | 30 (23-36) | 51 (41-60) | 59 (52-65) |
| % of patients progression-free at 12 months (95% CI) | 13 (8-20) | 22 (12-34) | 37 (29-45) |
| Objective response rate,a % (95% CI) | 36 (30-42) | 64 (55-72) | 63 (57-69) |
| Duration of responsea | |||
| No. of months, median (95% CI)b | 5.6 (4.2-9.6) | 6.9 (5.5-NE) | 9.4 (6.9-NE) |
| Intracranial progression-free survival | |||
| No. of months, median (95% CI) | 8.3 (7.3-11.3) | 12.5 (10.8-NE) | 12.8 (11.1-14.3) |
| % of patients progression-free at 6 months (95% CI) | 66 (59-72) | 78 (69-85) | 79 (74-84) |
| % of patients progression-free at 12 months (95% CI) | 34 (23-45) | 50 (35-64) | 54 (45-63) |
Efficacy analysis included all randomly assigned patients.
CI, confidence interval; NE, not estimable.
a No. of patients with measurable disease at baseline by blinded independent central review was 260 for chemotherapy, 130 for amivantamab–chemotherapy, and 259 for amivantamab–lazertinib–chemotherapy.
b Duration of response among confirmed responders.
At the time of this first interim overall survival analysis, the HRs for death were 0.77 (95% CI 0.49-1.21) for amivantamab–chemotherapy versus chemotherapy and 0.96 (95% CI 0.67-1.35) for amivantamab–lazertinib–chemotherapy versus chemotherapy (Supplementary Figure S9, available at https://doi.org/10.1016/j.annonc.2023.10.117).
Median intracranial PFS by blinded independent central review was 12.5 months (95% CI 10.8 months-not estimable) for amivantamab–chemotherapy, 12.8 months (95% CI 11.1-14.3 months) for amivantamab–lazertinib–chemotherapy, and 8.3 months (95% CI 7.3-11.3 months) for chemotherapy (Figure 3A; Table 2). The HR for intracranial disease progression or death was 0.55 (95% CI 0.38-0.79) for amivantamab–chemotherapy versus chemotherapy and 0.58 (95% CI 0.44-0.78) for amivantamab–lazertinib–chemotherapy versus chemotherapy. A sensitivity analysis was carried out among patients with a history of brain metastases and no prior brain radiotherapy (see Table 1 for subgroup). The intracranial PFS benefit of amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy was consistent with the overall population (Figure 3B). The median intracranial PFS was not estimable for amivantamab–chemotherapy (95% CI 5.6 months-not estimable) and 11.1 months for amivantamab–lazertinib–chemotherapy (95% CI 7.0-13.5 months) versus 6.3 months (95% CI 3.5-8.5 months) for chemotherapy, which corresponded to HRs for intracranial disease progression or death of 0.36 (95% CI 0.16-0.84) and 0.44 (95% CI 0.25-0.79), respectively.
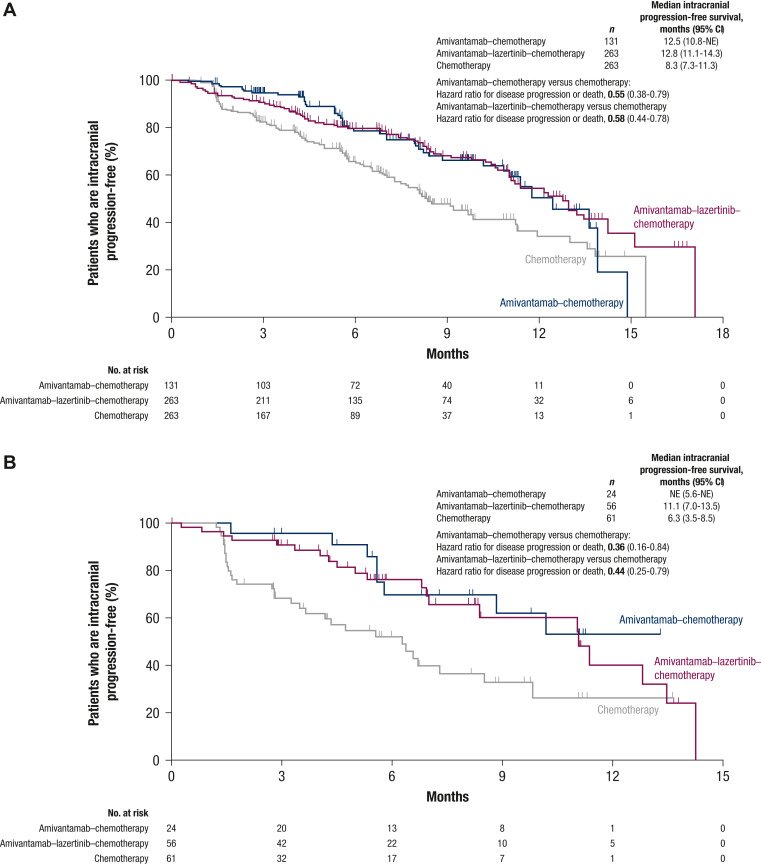
Safety
AEs of grade 3 or higher, mainly due to hematologic toxicities, were reported by 72% of patients treated with amivantamab–chemotherapy, 92% with amivantamab–lazertinib–chemotherapy, and 48% with chemotherapy (Table 3). The most common grade 3 or higher AEs (10% or higher in any arm) included neutropenia, thrombocytopenia, anemia, and leukopenia. Analysis of mean neutrophil and platelet counts over time revealed transient decreases during cycle 1 followed by recovery by cycle 2 day 1 and stabilization thereafter (Supplementary Figure S10, available at https://doi.org/10.1016/j.annonc.2023.10.117). The incidence of febrile neutropenia was 2%, 8%, and 2% for the amivantamab–chemotherapy, amivantamab–lazertinib–chemotherapy, and chemotherapy arms, respectively. Grade 3 or 4 bleeding events were seen in 1% of patients treated with amivantamab–chemotherapy, 3% with amivantamab–lazertinib–chemotherapy, and 0% with chemotherapy. Serious treatment-emergent AEs were observed in 32% of patients treated with amivantamab–chemotherapy, 52% with amivantamab–lazertinib–chemotherapy, and 20% with chemotherapy. The most common serious treatment-emergent AEs (5% or higher in any arm) were thrombocytopenia, neutropenia, and febrile neutropenia (Supplementary Table S3, available at https://doi.org/10.1016/j.annonc.2023.10.117).
Table 3 Treatment-emergent adverse events
| Event, n (%) | Chemotherapy (n = 243) | Amivantamab–chemotherapy (n = 130) | Amivantamab–lazertinib–chemotherapy (n = 263) |
| Any event | 227 (93) | 130 (100) | 263 (100) |
| Grade ≥3 | 117 (48) | 94 (72) | 242 (92) |
| Any serious event | 49 (20) | 42 (32) | 137 (52) |
| Any event resulting in death | 3 (1) | 3 (2) | 14 (5) |
| Any event leading to: | |||
| Interruptions of any study agent | 81 (33) | 84 (65) | 202 (77) |
| Reductions of any study agent | 37 (15) | 53 (41) | 171 (65) |
| Discontinuations of any study agent | 9 (4) | 24 (18) | 90 (34) |
| Adverse eventsa | All | Grade ≥3 | All |
| Neutropeniab | 101 (42) | 52 (21) | 74 (57) |
| Thrombocytopeniab | 72 (30) | 22 (9) | 57 (44) |
| Infusion-related reaction | 1 (0.4) | 0 | 76 (58) |
| Anemia | 97 (40) | 23 (9) | 51 (39) |
| Paronychia | 1 (0.4) | 0 | 48 (37) |
| Nausea | 90 (37) | 2 (1) | 58 (45) |
| Rash | 12 (5) | 0 | 56 (43) |
| Stomatitis | 21 (9) | 0 | 41 (32) |
| Leukopenia | 68 (28) | 23 (9) | 37 (28) |
| Hypoalbuminemia | 21 (9) | 1 (0.4) | 29 (22) |
| Constipation | 72 (30) | 0 | 50 (38) |
| Decreased appetite | 51 (21) | 3 (1) | 40 (31) |
| Peripheral edema | 15 (6) | 0 | 42 (32) |
| Vomiting | 42 (17) | 1 (0.4) | 32 (25) |
| Fatigue | 47 (19) | 4 (2) | 36 (28) |
| Diarrhea | 16 (7) | 1 (0.4) | 18 (14) |
| Asthenia | 40 (16) | 5 (2) | 34 (26) |
| Dermatitis acneiform | 7 (3) | 0 | 26 (20) |
| Alanine aminotransferase increased | 67 (28) | 10 (4) | 26 (20) |
| Hypokalemia | 15 (6) | 6 (2) | 24 (18) |
| COVID-19 | 25 (10) | 0 | 27 (21) |
| Hypocalcemia | 9 (4) | 0 | 16 (12) |
| Aspartate aminotransferase increased | 57 (23) | 0 | 19 (15) |
| Hyponatremia | 16 (7) | 2 (1) | 13 (10) |
| Pruritus | 17 (7) | 0 | 20 (15) |
| Adverse events of special interest | All | Grade ≥3 | All |
| Rashc | 30 (12) | 0 | 92 (71) |
| Venous thromboembolismd | 11 (5) | 7 (3) | 13 (10) |
| Interstitial lung diseasee | 0 | 0 | 2 (2) |
The safety population included all randomized patients who received at least one dose of any study treatment.
COVID-19, coronavirus disease 2019.
a Listed are adverse events by preferred term of all grades reported in ≥15% of patients in any treatment arm.
b For the amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy arms, ≥80% occurred in cycle 1 (within 21 days of treatment).
c Included the following preferred terms: rash, dermatitis acneiform, rash maculopapular, erythema, acne, rash pruritic, rash erythematous, rash macular, drug eruption, folliculitis, dermatitis, skin lesion, rash pustular, papule, rash follicular, exfoliative rash, pustule, rash papular, and skin exfoliation.
d Included the following preferred terms: pulmonary embolism, deep vein thrombosis, embolism, renal vein thrombosis, venous thrombosis limb, embolism venous, jugular vein thrombosis, superficial vein thrombosis, thrombophlebitis, and thrombosis.
e Included the following preferred terms: pneumonitis and interstitial lung disease.
Infusion-related reactions occurred in 58% and 56% of patients on amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy, respectively (Table 3). VTE occurred in 10%, 22%, and 5% of patients in the amivantamab–chemotherapy, amivantamab–lazertinib–chemotherapy, and chemotherapy arms, respectively (Table 3; Supplementary Table S4, available at https://doi.org/10.1016/j.annonc.2023.10.117). At the time of first VTE, very few patients were receiving anticoagulation (0% amivantamab–chemotherapy, 2% amivantamab–lazertinib–chemotherapy, 1% for chemotherapy). Treatment-related AEs are provided in Supplementary Table S5, available at https://doi.org/10.1016/j.annonc.2023.10.117.
Dose interruptions, reductions, and discontinuations due to AEs occurred in 84 (65%), 53 (41%), and 24 (18%) patients treated with amivantamab–chemotherapy, 202 (77%), 171 (65%), and 90 (34%) with amivantamab–lazertinib–chemotherapy, and 81 (33%), 37 (15%), and 9 (4%) with chemotherapy (Table 3). The most common reasons for interruptions, reductions, and discontinuations were hematologic toxicities (Supplementary Table S6, available at https://doi.org/10.1016/j.annonc.2023.10.117). Treatment-related AEs leading to discontinuation of all study agents were observed in 11 (8%), 25 (10%), and 6 (2%) patients receiving amivantamab–chemotherapy, amivantamab–lazertinib–chemotherapy, and chemotherapy, respectively.
Death within 30 days of the last dose occurred in 5%, 10%, and 3% of patients treated with amivantamab–chemotherapy, amivantamab–lazertinib–chemotherapy, and chemotherapy, respectively (Supplementary Table S7, available at https://doi.org/10.1016/j.annonc.2023.10.117; all grade 5 AEs in Supplementary Table S8, available at https://doi.org/10.1016/j.annonc.2023.10.117). Treatment-related AEs leading to death were infrequent in all arms; there were two (2%) deaths in the amivantamab–chemotherapy arm, four (2%) in the amivantamab–lazertinib–chemotherapy arm, and one (0.4%) in the chemotherapy arm. No clear pattern of specific AEs leading to death was detected.
Discussion
Amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy significantly improved PFS versus chemotherapy, with a 52% and 56% lower risk of disease progression or death, respectively. Early separation of curves was observed between both amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy versus chemotherapy. The PFS benefit was consistent across predefined subgroups. Mechanistically, amivantamab has shown efficacy against EGFR C797S, MET amplification, and other EGFR- and/or MET-based alterations after osimertinib.30 Chemotherapy provides activity against other resistance mechanisms that are EGFR- and MET-independent. Together, this combination provides broad coverage of the diverse and polyclonal tumor resistance arising after disease progression on osimertinib.It is notable that amivantamab–chemotherapy demonstrated similar intracranial PFS advantages over chemotherapy as amivantamab–lazertinib–chemotherapy. Amivantamab is a large molecule and was not expected to readily cross the blood–brain barrier. This was one of the key reasons for the addition of lazertinib, a known CNS-active TKI, to amivantamab–chemotherapy. The MARIPOSA-2 study shows that amivantamab–chemotherapy by itself can prevent or delay CNS recurrence. A similar improvement was seen among patients with a history of brain metastasis who had not received prior brain radiation, strengthening this conclusion. The mechanism by which amivantamab improves intracranial PFS could either be through direct antitumor effects or indirectly through immune-based mechanisms.Despite limited prospective data, third-generation TKIs are frequently continued after progression in combination with chemotherapy in an effort to mitigate development of CNS metastases.37 Data from this study and others indicate an elevated risk of cytopenic events when third-generation TKIs are administered concurrently with carboplatin.34,38, 39, 40 This toxicity necessitated a regimen change in the amivantamab–lazertinib–chemotherapy arm of this study. The rates of hematologic AEs were lower for amivantamab–chemotherapy versus amivantamab–lazertinib–chemotherapy. Notably, neutropenia was a leading cause of grade 3 or higher AEs for both amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy. Blood draws were carried out weekly in cycle 1, which captured transient decreases in neutrophil and platelet counts at the expected chemotherapy-induced nadir. Neutrophil and platelet counts were recovered by cycle 2 day 1 and stabilized. The rates of treatment discontinuations due to neutropenia and incidence of febrile neutropenia were low, further suggesting that the majority of neutropenia events were not clinically impactful. Consistent with prior reports, amivantamab-containing arms also had a higher incidence of EGFR- and MET-related AEs.33,41 The majority of these events were not serious (<2%) and of grade 1 or 2. In particular, discontinuations due to rash were infrequent, indicating rash was manageable through dose modifications and standard mitigation approaches. The incidences of infusion-related reactions in the amivantamab-containing arms were lower than reported for amivantamab monotherapy (56%-58% versus 67%). The majority were of grade 1-2 and did not lead to dose reductions or discontinuations. VTE rates were higher in patients receiving amivantamab–lazertinib–chemotherapy relative to the other arms, which is consistent with prior reports.35 There were no fatal VTEs, the events occurred early, and the majority were grade 1 or 2. It should be noted that despite the recommendation to use prophylactic anticoagulation for the first 4 months of treatment, utilization of anticoagulation was limited in MARIPOSA-2. The fact that the vast majority (>98%) of patients experiencing a venous thromboembolic event were not on anticoagulation, as well as the established safety and efficacy of anticoagulation in this population,42,43 implies that compliance with prophylactic anticoagulation is likely to mitigate this risk.Amivantamab–chemotherapy has a manageable toxicity profile, as noted by the low rates of discontinuations of all study agents due to treatment-related AEs. It should be noted that it is likely that the safety profile of amivantamab–lazertinib–chemotherapy will improve by not giving all four drugs simultaneously. Given the change to the amivantamab–lazertinib–chemotherapy regimen during the trial, more follow-up is required to rigorously characterize the safety and efficacy of the modified regimen.At present, there are no targeted therapies approved in the post-osimertinib setting. Two studies of immunotherapy–chemotherapy regimens have recently failed to show benefit over chemotherapy in the TKI-resistant setting.17,18 This highlights the unmet need in this patient population. Additionally, the ideal treatment in the second-line setting may further evolve as there are several novel regimens being evaluated in the first-line setting. At this time, however, amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy are the first regimens to demonstrate efficacy over chemotherapy in the post-osimertinib setting and could represent a new standard of care.MARIPOSA-2 did not require pretreatment tissue biopsies and instead collected blood samples for evaluating circulating tumor DNA (ctDNA). An NGS analysis of baseline ctDNA is planned; however, identification of ctDNA-based biomarkers has not previously been predictive of response.26,31 In addition, amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy improved clinical outcomes without the need for biomarker pre-selection.In summary, PFS and key secondary endpoints, such as objective response rate, duration of response, and intracranial PFS, were significantly improved with amivantamab–chemotherapy and amivantamab–lazertinib–chemotherapy compared with chemotherapy in patients with EGFR-mutated advanced NSCLC with disease progression on or after osimertinib.
Acknowledgements
The authors thank all the patients who participated in this study and their families and caregivers. The authors also thank the physicians and nurses who cared for the patients and the staff at the clinical sites. Medical writing assistance was provided by Katharine Fang, PhD (Janssen Global Services LLC), with support from Jessica Swanner, PhD and Jill Kolesar, PhD (Lumanity Communications Inc.) and funded by Janssen Global Services LLC.
Funding
This work was supported by Janssen Research & Development LLC. Medical writing assistance was funded by Janssen Global Services LLC. Funded by Janssen; MARIPOSA-2 (NCT04988295).
Disclosure
AP: consulting or advisory role for AstraZeneca, Bayer, Bristol Myers Squibb, Daiichi Sankyo, Lilly, GSK, Janssen, Merck Sharp & Dohme, Mundipharma, Novartis, and Roche; speakers bureaus for AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, eCancer, Janssen, Merck Sharp & Dohme, Medscape, PeerVoice, and touchONCOLOGY; steering committee member for Janssen and ArriVent Biopharma. SHL: received honoraria from AstraZeneca/MedImmune, Roche, Merck, Lilly, and Amgen; consulting or advisory role for AstraZeneca, Roche, Merck, Pfizer, and Lilly; received research funding from Merck. BM: received speaking honoraria from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, EMD Serono, Janssen, Jazz Pharmaceuticals, Merck, Novartis, Pfizer, Roche, Sanofi, and Takeda; received support for attending meetings from Janssen, Pfizer, and Sanofi. JYS: participated in advisory boards for AstraZeneca, Roche, Boehringer Ingelheim, Lilly, Pfizer, Novartis, Merck Sharp & Dohme, Chugai Pharmaceutical Co, Ono Pharmaceutical, Takeda, CStone Pharmaceuticals, Janssen, and Bristol Myers Squibb; received speaking honoraria from AstraZeneca, Roche, Boehringer Ingelheim, Lilly, Pfizer, Novartis, Merck Sharp & Dohme, Chugai Pharmaceutical Co, Ono Pharmaceutical, and Bristol Myers Squibb; received grant from Roche. KA: received lecture fees for AstraZeneca K.K., MSD K.K., Ono Pharmaceutical Co. Ltd., Bristol Myers Squibb, and Chugai Pharmaceutical Co. OJV: honoraria or advisory role for Bristol Myers Squibb, Merck Sharp & Dohme, Roche/Genentech, AstraZeneca, Pfizer, Lilly, Takeda, and Janssen; received travel, accommodations, and expenses from Takeda, AstraZeneca, Merck Sharp & Dohme, Pfizer, and Roche/Genentech. EF: consulting or advisory role for Amgen, AstraZeneca, Bayer, Bristol Myers Squibb, Lilly, GSK, Janssen, Merck Serono, Novartis, Pfizer, Sanofi, Takeda, Peptomyc, Daiichi Sankyo Europe GmbH, F. Hoffman La-Roche, Merck Sharp & Dohme, BerGenBio, and Turning Point Therapeutics; speakers bureaus for AstraZeneca, Bristol Myers Squibb, Lilly, Medscape, Merck Sharp & Dohme, PeerVoice, Pfizer, Takeda, Amgen, F. Hoffman La-Roche, Janssen, Medical Trends, Merck Serono, Sanofi, and touchONCOLOGY; other relationships with GRIFOLS; received research funding from Merck and Merck KgaA. NG: invited speaker for Amgen, AstraZeneca, Bristol Myers Squibb, Mirati Therapeutics, MSD, Novartis, Pfizer, Roche, and Sanofi; advisory roles for AbbVie, Amgen, AstraZeneca, Bristol Myers Squibb, Boehringer Ingelheim, Grünenthal, Janssen, Lilly, MSD, Novartis, Owkin, Pfizer, Roche, Sanofi, and Takeda; received research funding for institution from Bristol Myers Squibb, Janssen, Roche, and Sivan; leadership role at ITMIG; family employment at AstraZeneca. ABC: invited speaker for Pfizer, Amgen, Takeda, Novartis, Roche, AstraZeneca, MSD, Janssen, Bristol Myers Squibb, and Sanofi; advisory roles for Novartis, AbbVie, Roche, Exeliom Biosciences, Pfizer, Janssen, Amgen, Takeda, AstraZeneca, and MSD; research funding of institution from Exeliom Biosciences; support for attending meetings from Roche, MSD, Novartis, Pfizer, AstraZeneca, Amgen, and Bristol Myers Squibb; participation on data monitoring safety board for InhaTarget Therapeutics and Merck. RC: invited speaker for Amgen, AstraZeneca, Lilly, GSK, Janssen, MSD, Pfizer, Roche, and Takeda; participated in advisory boards for Amgen, AstraZeneca, Bayer, Lilly, GSK, Janssen, MSD, Novartis, Pfizer, PharmaMar, Roche, Sanofi, and Takeda; speaker in educational activities for Medscape, PeerVoice, and TouchIME; holds stocks or shares in Supportive Care UK; has ownership interest in Christie Private Care; principal investigator for AbbVie, AstraZeneca, Bristol Myers Squibb, Clovis, Lilly, GSK, Janssen, MSD, Novartis, Pfizer, PharmaMar, Roche, and Takeda. FC: received consulting fees from Roche, AstraZeneca, Bristol Myers Squibb, Pfizer, Takeda, Lilly, Bayer, Amgen, Sanofi, PharmaMar, Novocure, Mirati Therapeutics, Galecto, OSE Immunotherapeutics, and MSD; received honoraria for advisory role from Roche, AstraZeneca, Bristol Myers Squibb, Pfizer, Takeda, Lilly, Bayer, Amgen, Sanofi, PharmaMar, Novocure, Mirati Therapeutics, Galecto, OSE Immunotherapeutics, and MSD; received support for attending meetings from OSE Immunotherapeutics; participated in advisory boards for Roche, AstraZeneca, Bristol Myers Squibb, Pfizer, Takeda, Lilly, Bayer, Amgen, Sanofi, PharmaMar, Novocure, Mirati Therapeutics, Galecto, OSE Immunotherapeutics, and MSD. SO: received advisory board consulting fees from Janssen, Bristol Myers Squibb, Novocure, and Roche. SP: participated in advisory boards for Amgen, AstraZeneca, Bayer, BeiGene, Blueprint Medicines, Bristol Myers Squibb, Boehringer Ingelheim, Daiichi Sankyo, EQRx, GSK, Guardant Health, Janssen, Lilly, Medscape, MSD, Novartis, Pfizer, Sanofi, and Takeda; consulting fees from Amgen, AstraZeneca, Bayer, Blueprint Medicines, Bristol Myers Squibb, Boehringer Ingelheim, Daiichi Sankyo, GSK, Guardant Health, Incyte, Janssen, Lilly, Merck Serono, MSD, Novartis, Roche, Takeda, Pfizer, Seattle Genetics, Turning Point Therapeutics, and EQRx; expert testimony for Merck Serono and Roche; invited speaker for VJOncology; received payment or honoraria from AstraZeneca, Bayer, Guardant Health, Janssen, Merck Serono, Roche, and Takeda; principal investigator for ARIAD Pharmaceuticals, AstraZeneca, Bristol Myers Squibb, Boehringer Ingelheim, Celgene, Daiichi Sankyo, Janssen, Lilly, Roche, Takeda, Trizel, and Turning Point Therapeutics; received support for attending meetings from Janssen and Roche; research grant from Guardant Health; other financial relationships with Amgen, Blueprint Medicines, Elsevier, and MSD. JLT: invited speaker for AstraZeneca, Lilly, MSD, Boehringer Ingelheim, Pfizer, and Takeda; participated in advisory boards for Amgen and AstraZeneca; received support for attending meetings from MSD. PT: received grants and nonfinancial support from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Novartis, Roche, and Takeda. RDG: received grants from Alliance Foundation, Amgen, AstraZeneca, Big Ten Cancer Research Consortium, Bristol Myers Squibb, Chugai Pharmaceutical Co, Daiichi Sankyo, ECOG-ACRIN, Helsinn, Hoosier Cancer Research Network, Janssen, Jounce Therapeutics, Merck, National Cancer Institute, Pfizer, and Takeda; received payment or honoraria from Clinical Care Options, OncLive, Society for Immunotherapy of Cancer, and Targeted Oncology; received support for attending meetings from the International Association for the Study of Lung Cancer; participated in advisory boards for AstraZeneca, Blueprint Medicines, Daiichi Sankyo, Gilead, Janssen, Jazz Pharmaceuticals, Mirati Therapeutics, Oncocyte, Sanofi, and Takeda; leadership or other role in ASCO Scientific Review Committee and Meeting Abstracts, Hoosier Cancer Research Network, Journal of Clinical Oncology, National Cancer Institute Investigational Drug Steering Committee, and the Thoracic Clinical Trial Working Group. WNWJr: participated in advisory boards for AstraZeneca, Bayer, Merck, Novartis, Sanofi, and Takeda; invited speaker for Amgen, Bristol Myers Squibb, Lilly, Genentech/Roche, Janssen, Pfizer, and United Medical; expert testimony for Boehringer Ingelheim. KR: consultant for Amgen, AstraZeneca, Blueprint Medicines, Daiichi Sankyo, EMD Serono, Genentech, GSK, Janssen, Lilly, Merck KGA, Mirati Therapeutics, Seattle Genetics, and Takeda; writing assistance from Blueprint Medicines, Genentech, Janssen, Merck, Mirati Therapeutics, Seattle Genetics, and Takeda; research funding of institution from Genentech, Blueprint Medicines, Calithera, Daiichi Sankyo, Elevation Oncology, and Janssen. TT: received honoraria from AstraZeneca K.K., Pfizer Japan Inc, Eli Lilly Japan, Chugai Pharmaceutical Co, MSD K.K., Pfizer Japan Inc, Takeda, Bristol Myers Squibb Japan, Amgen K.K., and Novartis; research funding of institution from Janssen Pharmaceutical K.K., AstraZeneca K.K., Pfizer Japan Inc, Eli Lilly Japan, Chugai Pharmaceutical Co, MSD K.K., Amgen K.K., Merck Biopharma Co., Ltd., and AnHeart Therapeutics Inc. SG: principal investigator for AstraZeneca, Novartis, and Janssen (fees to institution). DMK: participated in advisory boards for MSD, Merck, Roche, Bristol Myers Squibb, Takeda, Boehringer Ingelheim, Pfizer, Johnson & Johnson, Amgen, Sanofi-Aventis, and Novartis. AB: received consulting fees from Pfizer and Roche; honoraria from Novartis and Eli Lilly; participated in advisory boards for Pfizer, Roche, and Regeneron. MM: received consulting fees from Boehringer Ingelheim, Roche, Takeda, and AstraZeneca. PB, ABB, AG, JG, DM, MW, JX, TS, SS, BD, REK, JMB: employee of Janssen and may hold stock in Johnson & Johnson. RGC: invited speaker for AstraZeneca, Bristol Myers Squibb, Janssen, Lilly, Novartis, Pfizer, Roche, and Takeda; advisory roles for AstraZeneca, Bristol Myers Squibb, Janssen, Lilly, MSD, Novartis, Pfizer, Roche, Sanofi, and Takeda; steering committee member for AstraZeneca and Janssen. BCC: consulting or advisory role for AstraZeneca, Boehringer Ingelheim, Roche, Bristol Myers Squibb, Pfizer, Yuhan Corporation, Janssen, Takeda, Merck Sharp & Dohme, Ono Pharmaceutical, Lilly, MedPacto, Blueprint Medicines, Cyrus Therapeutics, Guardant Health, Novartis, CJ, ABION, BeiGene, CureLogen, Onegene Biotechnology, GI-Cell, HK inno.N, IMNEWRUN, Hanmi Pharmaceutical, Kanaph Therapeutics, BridgeBio, and Oscotec; leadership roles for Interpark Bio and J INTS BIO; patents, royalties, or other intellectual property for Champions Oncology, Crown Bioscience, and Imagen; other relationships with DAAN Biotherapeutics; owns stock or has other ownership interests with Theravance Biopharma, Gencurix, BridgeBio, Kanaph Therapeutics, Cyrus Therapeutics, Interpark Bio, and J INTS BIO; received research funding from Novartis, Bayer, AstraZeneca, MOGAM Biotechnology Research Institute, Dong-A ST, Champions Oncology, Janssen, Yuhan Corporation, Ono Pharmaceutical, Dizal Pharma, Merck Sharp & Dohme, AbbVie, GI Innovation, Lilly, Blueprint Medicines, Interpark Bio, LG Chem, Oscotec, GI-Cell, ABION, Boehringer Ingelheim, CJ Bioscience, CJ Blossom Park, Cyrus Therapeutics, Genexine, Nuvalent Inc, Oncternal Therapeutics, Regeneron, BridgeBio, ImmuneOncia, Illumina, Kanaph Therapeutics, Therapex, J INTS Bio, Hanmi Pharmaceutical, and CHA Bundang Medical Center. All other authors have declared no conflicts of interest.
Data sharing
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinicaltrials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at http://yoda.yale.edu.
Supplementary data
References
- RYBREVANT. RYBREVANT (amivantamab-vmjw) Injection, for Intravenous Use [prescribing information]. Janssen Biotech, Inc, Horsham, PA2021View in Article
- Google Scholar
- Bauml J.
- Cho B.C.
- Park K.
- et al.
- Amivantamab in combination with lazertinib for the treatment of osimertinib-relapsed, chemotherapy-naïve EGFR mutant (EGFRm) non-small cell lung cancer (NSCLC) and potential biomarkers for response. Paper presented at the American Society of Clinical Oncology Annual Meeting.June 4-8, 2021View in Article
- Google Scholar
- Haura E.B.
- Cho B.C.
- Lee J.S.
- et al.
- JNJ-61186372 (JNJ-372), an EGFR-cMet bispecific antibody, in EGFR-driven advanced non-small cell lung cancer (NSCLC).J Clin Oncol. 2019; 37: 9009View in Article
- Google Scholar
- Krebs M.
- Spira A.I.
- Cho B.C.
- et al.
- Amivantamab in patients with NSCLC with MET exon 14 skipping mutation: updated results from the CHRYSALIS study.J Clin Oncol. 2022; 40: 9008View in Article
- Google Scholar
- Cho B.C.
- Kim D.-W.
- Spira A.I.
- et al.
- Amivantamab plus lazertinib in osimertinib-relapsed EGFR-mutant advanced non-small cell lung cancer: a phase 1 trial.Nat Med. 2023; https://doi.org/10.1038/s41591-023-02554-7View in Article
- Google Scholar
- Yun J.
- Hong M.H.
- Kim S.Y.
- et al.
- YH25448, an irreversible EGFR-TKI with potent intracranial activity in EGFR mutant non–small cell lung cancer.Clin Cancer Res. 2019; 25: 2575-2587View in Article
- Google Scholar
- Ahn M.J.
- Han J.Y.
- Lee K.H.
- et al.
- Lazertinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: results from the dose escalation and dose expansion parts of a first-in-human, open-label, multicentre, phase 1-2 study.Lancet Oncol. 2019; 20: 1681-1690View in Article
- Google Scholar
- Shu C.A.
- Goto K.
- Ohe Y.
- et al.
- Amivantamab and lazertinib in patients with EGFR-mutant non-small cell lung cancer (NSCLC) after progression on osimertinib and platinum-based chemotherapy: updated results from CHRYSALIS-2. Paper presented at the 58th Annual Meeting of the American Society of Clinical Oncology (ASCO), Chicago, ILJune 3-7, 2022View in Article
- Google Scholar
- Leighl N.B.
- Shu C.A.
- Minchom A.
- et al.
- 1192MO Amivantamab monotherapy and in combination with lazertinib in post-osimertinib EGFR-mutant NSCLC: analysis from the CHRYSALIS study.Ann Oncol. 2021; 32: S951-S952View in Article
- Google Scholar
- Besse B.
- Baik C.S.
- Marmarelis M.E.
- et al.
- Predictive biomarkers for treatment with amivantamab plus lazertinib among EGFR-mutated NSCLC in the post-osimertinib setting: analysis of tissue IHC and ctDNA NGS.J Clin Oncol. 2023; 41 (Paper presented at the 59th Annual Meeting of the American Society of Clinical Oncology (ASCO). June 2-6, 2023; Chicago, IL): 9013View in Article
- Google Scholar
- White M.N.
- Piotrowska Z.
- Stirling K.
- et al.
- Combining osimertinib with chemotherapy in EGFR-mutant NSCLC at progression.Clin Lung Cancer. 2021; 22: 201-209View in Article
- Google Scholar
- Nagasaka M.
- Goto K.
- Gomez J.
- et al.
- P50.04 Amivantamab in combination with chemotherapy in patients with advanced non-small cell lung cancer (NSCLC).J Thorac Oncol. 2021; (ePoster presented at the 2021 World Conference on Lung Cancer; September 8, 2021; Worldwide Virtual Event 2021)16:S1116View in Article
- Google Scholar
- Marmarelis M.
- Lee S.H.
- Spira A.I.
- et al.
- Amivantamab and lazertinib in combination with platinum-based chemotherapy in relapsed/refractory EGFR-mutant NSCLC. Oral presented at the World Conference on Lung Cancer 2022. Vienna, AustriaAugust 6-9, 2022View in Article
- Google Scholar
- Girard N, Cho BC, Spira AI, et al. Risk factors for venous thromboembolism (VTE) among patients with EGFR-mutated advanced non-small cell lung cancer (NSCLC) receiving amivantamab plus lazertinib versus either agent alone. Paper presented at the American Society of Oncology (ASCO) annual meeting; June 2-6, 2023; Chicago, IL, USA.
View in Article- Google Scholar
- Eisenhauer E.A.
- Therasse P.
- Bogaerts J.
- et al.
- New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1).Eur J Cancer. 2009; 45: 228-247View in Article
- Google Scholar
- Goulart B.H.L.
- Larkins E.
- Beaver J.A.
- Singh H.
- Continuation of third-generation tyrosine kinase inhibitors in second-line trials for EGFR-mutated non-small-cell lung cancer: regulatory considerations.J Clin Oncol. 2023; 41: 3905-3908View in Article
- Google Scholar
- Li Y.
- Pan Z.Y.
- Zhang Y.
- Li L.
- EP08.02-003 Aumolertinib plus chemotherapy as 1st line treatment in advanced lung cancer EGFR mutation and ctDNA cleared analysis.J Thorac Oncol. 2022; 17: S397View in Article
- Google Scholar
- Nakamura A.
- Saito R.
- Ko R.
- et al.
- A phase II study of osimertinib in combination with platinum plus pemetrexed in patients with EGFR-mutated, advanced non–small cell lung cancer: the OPAL study (NEJ032C/LOGIK1801).J Clin Oncol. 2022; 40 (9097): 9097View in Article
- Google Scholar
- Tanaka K.
- Asahina H.
- Kishimoto J.
- et al.
- Osimertinib versus osimertinib plus chemotherapy for non-small cell lung cancer with EGFR (T790M)-associated resistance to initial EGFR inhibitor treatment: an open-label, randomised phase 2 clinical trial.Eur J Cancer. 2021; 149: 14-22View in Article
- Google Scholar
- Lee S.
- Cho B.
- Spira A.
- et al.
- Amivantamab, lazertinib plus platinum-based chemotherapy in EGFR-mutated advanced NSCLC: updated results from CHRYSALIS-2. Paper presented at the 2023 World Conference on Lung Cancer (WCLC).September. 2023; (Singapore): 9-12View in Article
- Google Scholar
- Zhang J.
- Zhang Y.L.
- Ma K.X.
- Qu J.M.
- Efficacy and safety of adjunctive anticoagulation in patients with lung cancer without indication for anticoagulants: a systematic review and meta-analysis.Thorax. 2013; 68: 442-450View in Article
- Google Scholar
- Lee J.H.
- Hyun D.G.
- Choi C.M.
- et al.
- A retrospective study on efficacy and safety of rivaroxaban and dalteparin for long-term treatment of venous thromboembolism in patients with lung cancer.Respiration. 2019; 98: 203-211View in Article
- Google Scholar
Article info
Publication history
Published online: October 23, 2023
Identification
DOI: https://doi.org/10.1016/j.annonc.2023.10.117
Copyright
© 2023 The Author(s). Published by Elsevier Ltd on behalf of European Society for Medical Oncology.
User license
Creative Commons Attribution – NonCommercial – NoDerivs (CC BY-NC-ND 4.0) | How you can reuse
ScienceDirect
Access this article on ScienceDirect
Figures
- Graphical Abstract
- Figure 1Progression-free survival by blinded independent central review and by investigator.
- Figure 2Progression-free survival by blinded independent central review of patient subgroups.
- Figure 3Intracranial progression-free survival.
Tables
- Table 1Demographics and baseline disease characteristics
- Table 2Key efficacy endpoints by blinded independent central review
- Table 3Treatment-emergent adverse events
Linked Article
- POSing the question: MARIPOSA-2, do the ends justify the means?Annals of OncologyVol. 35Issue 1


