FDA grants accelerated approval to tarlatamab-dlle for extensive stage small cell lung cancer
What is the potential role for tarlatamab-dlle in the treatment of small cell lung cancer?
- Tarlatamab-dlle is a bispecific T-Cell Engager (BsAbs) that binds to Delta-like ligand 3 (DLL3) expressed on the small cell lung cancer (SCLC) tumor cell surface with one arm and to CD3 on T-Cells with the other arm, creating cytolytic synapse that leads to apoptosis of SCLC. 1
- Tarlatamab-dlle is recommended in the National Comprehensive Cancer Network (NCCN) guidelines as a second line treatment option for ES-SCLC in patients with either relapse or primary progressive disease.
- Approval for this indication was based on the DeLLphi-301 trial, an open-label, multicenter, multi-cohort, phase II study in adults with recurrent or progressive SCLC who were previously treated with two or more lines of therapy.2
- Patients 18 years of age or older, with Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, and progression or recurrence of disease following one platinum-based regimen and at least one other prior line of therapy, were treated with tarlatamab-dlle 1 mg on day 1 and 10 mg on days 8, 15, and every 2 weeks thereafter.
- Table 1: Results of patients who received at least 1 dose of tarlatamab-dlle (n=99)
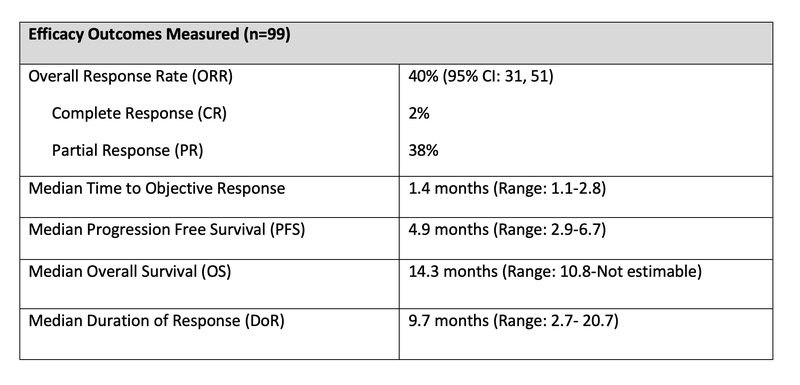
- The most common adverse reactions observed were cytokine release syndrome (CRS) (55%), with 1.6% being grade 3 or higher. In the pooled safety population, 47% of patients experienced neurologic toxicity, including 9% cases of immune effector cell-associated neurotoxicity (ICANS).1,2
- Other treatment options that can be used second line in ES-SCLC include topotecan, lurbinectedin and irinotecan.3,5
- These options usually have short DoR (3.6-5.3 months) and OS that is around 8 months.2
What role can the pharmacist play in the management of patients on tarlatamab-dlle?
- Pharmacists play essential roles in managing the correct dosage, frequency, and scheduling of each step of the treatment process to ensure efficacy and safety.1
- Recommended Tarlatamab dosing and monitoring schedule (28-day treatment cycles):
Table 2: Tarlatamab-dlle dosing schedule
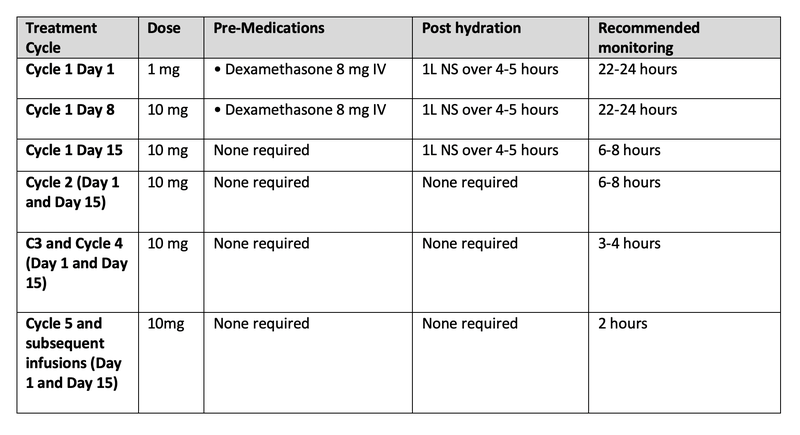
- Pharmacists play a critical role in monitoring and managing early adverse events associated with tarlatamab-dlle, such as CRS, neurotoxicity, and ICANS. They are instrumental in guiding dose adjustments based on the severity of these toxicities.
- The most common (>20%) adverse reactions were CRS (55%), fatigue (51%), neurologic toxicity including ICANS (47%), pyrexia (36%), dysgeusia (36%), decreased appetite (34%), musculoskeletal pain (30%), constipation (30%), anemia (27%) and nausea (22%).
- Pharmacists must be well-versed in the management of these adverse events and actively contribute to the development and ongoing refinement of institutional protocols and operational procedures.
- CRS prevention and management are major areas pharmacists can play a key role.1
- Pharmacists should be looking out for signs and symptoms of fever, hypotension, hypoxia, nausea, vomiting, fatigue, tachycardia and headache. The median time to onset of all grade CRS was 13.5 hours (Range: 1 to 268 hours). CRS is most likely to occur following the first two dose of tarlatamab-dlle, given on days 1 and 8 of cycle 1.
- CRS prevention:
- Ensure that correct frequency and schedule is ordered for step-up dosing. For gaps in therapy, an abbreviated dose ramp-up may need to be repeated.
- Patients should be counseled on signs and symptoms of CRS and when to seek medical attention.
- CRS management:
- For Grade 1-2 CRS, hold tarlatamab-dlle until event resolves, then resume at the next scheduled dose. Administer symptomatic treatments if needed.
- For Grade 2 CRS, consider administration of dexamethasone or tocilizumab. When resuming treatment, monitor patients from the start of the infusion for 22-24 hours.
- For Grade 3 CRS, hold tarlatamab-dlle until the event resolves, then resume at the next scheduled dose. In addition to symptomatic treatments, recommend administering tocilizumab and monitoring in the appropriate hospital setting.
- When resuming treatment, administer concomitant medications as recommended for cycle 1. Monitor patients for 22-24 hours.
- For Grade 4 CRS, permanently discontinue tarlatamab-dlle.
- ICANS is a serious or life-threatening neurologic toxicity that can occur in patients receiving tarlatamab-dlle therapy, and pharmacists can make a key impact in prevention and management of ICANS based on ICE Score.1
- Pharmacists should be looking out for signs and symptoms of confusion, impaired motor skills and word finding difficulties. The median time to onset of ICANS from the first dose was 29.5 days (range 1-154 days). ICANS and associated neurologic events occurred most following Cycle 2 Day 1.
- ICANS prevention:
- Patients should be counseled in the signs and symptoms of ICANS and when to seek medical attention.
- ICANS management:
- For Grade 1-2 ICANS, hold tarlatamab-dlle until event resolves, then resume at the next scheduled dose. Administer supportive treatments if needed.
- For Grade 2 ICANS, consider administration of dexamethasone if symptoms worsen. When resuming treatment, monitor patients from the start of the infusion for 22-24 hours.
- For Grade 3 ICANS, hold tarlatamab-dlle until the event resolves, then resume at the next scheduled dose. If there is no improvement within 7 days or grade 3 toxicity reoccurs within 7 days of reinitiation, permanently discontinue.
- Recommend monitoring in the appropriate hospital setting and administrating dexamethasone. Monitor patients for 22-24 hours upon reinitiation.
- For Grade 4 ICANS, permanently discontinue tarlatamb-dlle. Recommend high-dose corticosteroids.
Clinical Pearls
- Step-up dosing is required to initiate tarlatamab-dlle to minimize the risk and severity of CRS, neurotoxicity, and ICANS. 1
- Tarlatamab-dlle is currently under accelerated approval by the FDA and is not part of a REMS program.
- Based on its mechanism of action, it may pose a potential risk to a fetus. Females of reproductive potential should be advised to use effective contraception during treatment and for 2 months following the last dose.
- Administer the reconstituted and diluted tarlatamab-dlle over 1 hour at a constant flow rate of 250mL/hr.1
- Tarlatamab-dlle can be stored in a refrigerator at 2°C to 8°C (36°F to 46°F) for up to 24 hours in the original carton to protect from light.
- The prepared tarlatamab-dlle can be stored in the room temperature (20°C to 25°C for up to 8 hours and in the refrigerated temperature (2°C to 8°C) for up to 7 days.
- Tarlatamab-dlle is the first BsAbs herapy targeting DLL3 to gain approval SCLC through DeLLphi-301 trial.
- It is important to note that DeLLphi-301 trial excluded patients with untreated or symptomatic brain metastasis and leptomeningeal disease.2
- Currently, the DeLLphi-304 trial is underway, a randomized, open-label, Phase III study comparing tarlatamab-dlle to standard treatments in patients with relapsed SCLC following platinum-based chemotherapy. The standard treatments included in this trial are lurbinectedin, topotecan, and amrubicin.7
References
1.Tarlartamab-dlle (Imdelltra) [prescribing information]. Thousand Oaks, CA: Amgen Inc; 2024.
2.Ahn MJ et al. DeLLphi-301 Investigators. Tarlatamab for Patients with Previously Treated Small-Cell Lung Cancer. N Engl J Med. 2023 Nov 30;389(22):2063-2075. doi: 10.1056/NEJMoa2307980. Epub 2023 Oct 20. PMID: 37861218.
3.National Comprehensive Cancer Network. Small Cell Lung Cancer (Version 1.2025). Updatde September 5, 2024. Accessed September 12, 2024. https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf.
4.Morgensztern D et al. Efficacy and Safety of Rovalpituzumab Tesirine in Third-Line and Beyond Patients with DLL3-Expressing, Relapsed/Refractory Small-Cell Lung Cancer: Results From the Phase II TRINITY Study. Clin Cancer Res. 2019 Dec 1;25(23):6958-6966. doi: 10.1158/1078-0432.CCR-19-1133. Epub 2019 Sep 10. PMID: 31506387; PMCID: PMC7105795
5.Blackhall F et al. Efficacy and Safety of Rovalpituzumab Tesirine Compared With Topotecan as Second-Line Therapy in DLL3-High SCLC: Results From the Phase 3 TAHOE Study. J Thorac Oncol. 2021 Sep;16(9):1547-1558. doi: 10.1016/j.jtho.2021.02.009. Epub 2021 Feb 16. PMID: 33607312.
6.Johnson ML et al. Rovalpituzumab Tesirine as a Maintenance Therapy After First-Line Platinum-Based Chemotherapy in Patients With Extensive-Stage-SCLC: Results From the Phase 3 MERU Study. J Thorac Oncol. 2021 Sep;16(9):1570-1581. doi: 10.1016/j.jtho.2021.03.012. Epub 2021 Apr 3. PMID: 33823285.
7.Luis G. Paz-Ares et al. Randomized phase 3 study of tarlatamab, a DLL3-targeting bispecific T-cell engager (BiTE), compared to standard of care in patients with relapsed small cell lung cancer (DeLLphi-304).. JCO 41, TPS8611-TPS8611(2023) DOI:10.1200/JCO.2023.41.16_suppl.TPS8611




